

MagReSyn® Amine
Primary Amine functional support for ligand adsorption or chemical modification
Amine (NH2) functional microparticles may be used to physically adsorb biomolecules through ionic interaction, or they can be chemically functionalized for covalent coupling of biomolecules using a variety of bi-functional chemical coupling agents. The most frequently used coupling agents include glutaraldehyde and 1,4-butanediol diglycidyl ether.
For possible coupling chemistries and protocols we recommend you consult with the following reference: Hermanson, G.T. Bioconjugate Techniques, 2nd Edition, Academic Press Inc. (2008)
Support: Proprietary polymer microparticles containing iron oxide (magnetite)
Binding capacity: > 20 mg.ml-1 (BSA)
Bead size: ~5-10 µm
Formulation: 20 mg.ml-1 suspension in 20% ethanol
Product Resources
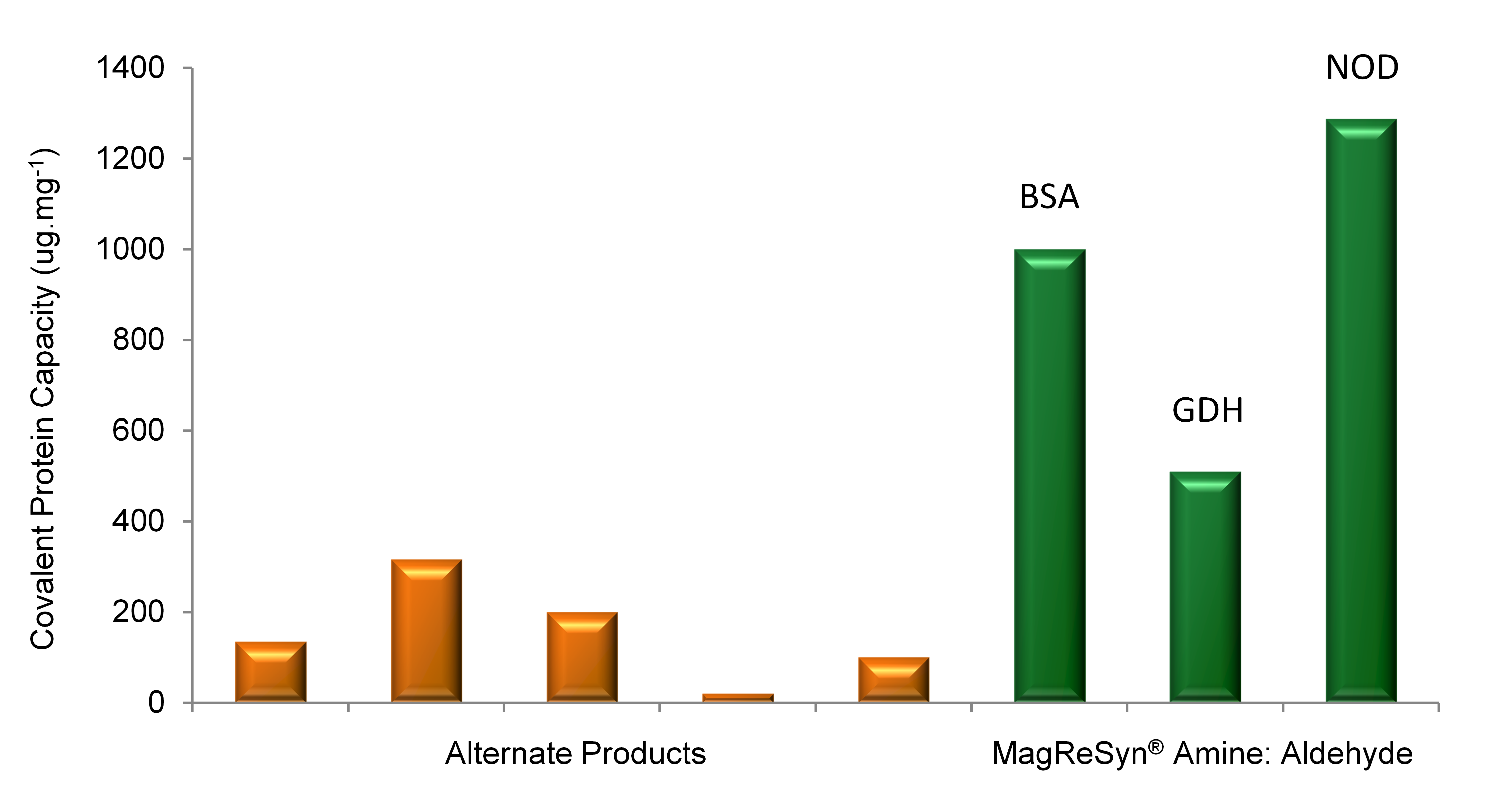
MagReSyn® Amine Performance Superiority
Aldehyde-activated
Epoxide-activated

TERMS AND CONDITIONS
Products supplied by ReSyn Biosciences (Pty) Ltd are for research purposes only. ReSyn products are not to be used for diagnostic, therapeutic or commercial means any use resulting in monetary gain, including, but not limited to, incorporation in a kit, repackaging and re-formulation. Please enquire about sub-licenses for commercial use.


